Gay-Lussac LAw

Gay-Lussac was born at Saint-Leonard-de-Noblat in the present-day department of Haute-Vienne. He received his early education at the hands of the Catholic Abbey of Bourdeix. In 1831 he was elected to represent Haute-Vienne in the chamber of deputies, and in 1839 he entered the chamber of peers.
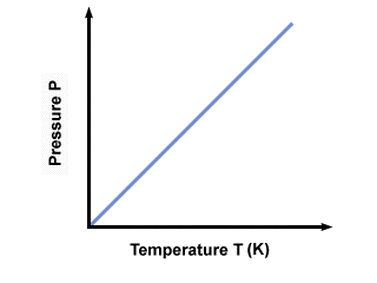
He first formulated the Gay-Lussac Law stating that if the mass and volume of a gas are held constant then gas pressure increases linearly as the temperature rises. This is sometimes written as P=k T, where k is a constant dependent on the mass and volume of the gas and T is temperature on an absolute scale.
He first formulated the Gay-Lussac Law stating that if the mass and volume of a gas are held constant then gas pressure increases linearly as the temperature rises. This is sometimes written as P=k T, where k is a constant dependent on the mass and volume of the gas and T is temperature on an absolute scale.

Gay-Lussac's law states that p1/t1=p2/t2 under constant volume. The variables can be defined as follows: p1 is initial pressure, t1 is initial temperature, p2 is final pressure, and t2 is final temperature. If temperature increases then pressure also increases. This explains why on a hot day a plastic bottle of pop left in vehicle can feel very stiff, because of increased pressure in the bottle due to elevated temperature.
Someone opening an oven may feel a quick flow of hot air; the air inside the oven is heated, therefore pressurized. The same is true when heating food in closed containers; often, a container will open to release the pressure. If it does not, opening the container will quickly release all the pent-up pressure, which can be very dangerous because the gases inside the hot container may be super-heated. This is why it is always best to open hot containers away from your body and face.
Someone opening an oven may feel a quick flow of hot air; the air inside the oven is heated, therefore pressurized. The same is true when heating food in closed containers; often, a container will open to release the pressure. If it does not, opening the container will quickly release all the pent-up pressure, which can be very dangerous because the gases inside the hot container may be super-heated. This is why it is always best to open hot containers away from your body and face.
Practice Problems
1. Problem:
A 20 L cylinder containing 6 atm of gas at 27 °C. What would the pressure of the gas be if the gas was heated to 77 °C?
Solution
The cylinder's volume remains unchanged while the gas is heated so Gay-Lussac's gas law applies. Gay-Lussac's gas law can be expressed as P1/T1 = P2/T2 where P1 and T1 are the initial pressure and absolute temperatures P2 and T2 are the final pressure and absolute temperature
First, convert the temperatures to absolute temperatures.
T1 = 27 °C = 27 + 273 K = 300 K
T2 = 77 °C = 77 + 273 K = 350 K
Use these values in Gay-Lussac's equation and solve for P2.
P2 = PiTf/Ti
P2 = (6 atm)x(350K)/(300 K)
P2 = 7 atm
Answer:
The pressure will increase to 7 atm after heating the gas from 27 °C to 77 °C.
2.Problem:
A sample of hydrogen at 46◦C exerts a pres-
sure of 0.483 atm. The gas is heated to 76◦C atconstant volume. What will its new pressure
be?
p1 / T1 = p2 / T2
p: pressure
T: temperature (in Kelvin!)
1: starting conditions
2: end conditions.
You need to calculate p2. So the equation can be changed to:
p1 / T1 * T2 = p2
Inserting the values:
0.483 / (273 + 46) * (273 + 76) = 0.483 / 319 * 349 = 0.53 atm
3. Problem
9.0 L of a gas is found to exert 83.0 kPa at 35.0°C. What would be the required temperature (in Celsius) to change the pressure to standard pressure?
Answer:
We must first change 35.0°C to 308.0 K and remember that standard pressure in the kiloPascal unit (kPa) is 101.325. Insert values into the equation, giving us:
Answer:
375.9 °K, but the question asks for Celsius, so you subtract 273 to get the final answer of 102.9 °C.
4.Problem:
The pressure of a container of helium is 650 torr. If the sealed container is cooled to 0oC, what will the pressure be?
Solution:
Since the container is sealed, the volume does not change. Remember that temperature must be in kelvins.
P1 = 650 torr; T1 = 25oC = 298K; T2 = 0oC = 273K
Solving for P2
Since the problem is concerned only with temperature and pressure.
P2 = [650 torr][273K]298K = 595 torr
5.Problem:
If we have a tank of gas at 1.520 torr pressure and a temperature of 300K and it is heated to 500K. What is the new pressure in atmospheres?
Solution:
Convert the initial pressure to atmospheres.
1520 torr1 × 1 atm760 torr = 2.00atm
Solve for P2 in Gay-Lussac's law
P2 = P1T2T1
Substitute the values
P2 = [2.00 atm][500K]300K = 3.33atm.
A 20 L cylinder containing 6 atm of gas at 27 °C. What would the pressure of the gas be if the gas was heated to 77 °C?
Solution
The cylinder's volume remains unchanged while the gas is heated so Gay-Lussac's gas law applies. Gay-Lussac's gas law can be expressed as P1/T1 = P2/T2 where P1 and T1 are the initial pressure and absolute temperatures P2 and T2 are the final pressure and absolute temperature
First, convert the temperatures to absolute temperatures.
T1 = 27 °C = 27 + 273 K = 300 K
T2 = 77 °C = 77 + 273 K = 350 K
Use these values in Gay-Lussac's equation and solve for P2.
P2 = PiTf/Ti
P2 = (6 atm)x(350K)/(300 K)
P2 = 7 atm
Answer:
The pressure will increase to 7 atm after heating the gas from 27 °C to 77 °C.
2.Problem:
A sample of hydrogen at 46◦C exerts a pres-
sure of 0.483 atm. The gas is heated to 76◦C atconstant volume. What will its new pressure
be?
p1 / T1 = p2 / T2
p: pressure
T: temperature (in Kelvin!)
1: starting conditions
2: end conditions.
You need to calculate p2. So the equation can be changed to:
p1 / T1 * T2 = p2
Inserting the values:
0.483 / (273 + 46) * (273 + 76) = 0.483 / 319 * 349 = 0.53 atm
3. Problem
9.0 L of a gas is found to exert 83.0 kPa at 35.0°C. What would be the required temperature (in Celsius) to change the pressure to standard pressure?
Answer:
We must first change 35.0°C to 308.0 K and remember that standard pressure in the kiloPascal unit (kPa) is 101.325. Insert values into the equation, giving us:
Answer:
375.9 °K, but the question asks for Celsius, so you subtract 273 to get the final answer of 102.9 °C.
4.Problem:
The pressure of a container of helium is 650 torr. If the sealed container is cooled to 0oC, what will the pressure be?
Solution:
Since the container is sealed, the volume does not change. Remember that temperature must be in kelvins.
P1 = 650 torr; T1 = 25oC = 298K; T2 = 0oC = 273K
Solving for P2
Since the problem is concerned only with temperature and pressure.
P2 = [650 torr][273K]298K = 595 torr
5.Problem:
If we have a tank of gas at 1.520 torr pressure and a temperature of 300K and it is heated to 500K. What is the new pressure in atmospheres?
Solution:
Convert the initial pressure to atmospheres.
1520 torr1 × 1 atm760 torr = 2.00atm
Solve for P2 in Gay-Lussac's law
P2 = P1T2T1
Substitute the values
P2 = [2.00 atm][500K]300K = 3.33atm.