Solutions
A solution is a liquid mixture in which the minor component (the solute) is uniformly distributed within the major component (the solvent).

- Chocolate milk is an example of solution; 2 spoons of chocolate syrup would be the solvent, and 2 cups of milk is the solute.
- Factors that affect the rate of solution is temperature, them amount of pressure, and the amount of solute or solvent used.
- temperature, pressure, and polarity affect the solubility. Gaseous solutes exhibit more complex behavior with temperature. As the temperature is raised gases usually become less soluble in water, but more soluble in organic solvents.
MOlarity
Molarity=moles solute divided by liters of solution
- H ow many grams of CaCl2 are in 350 ml of 2.0M CaCl2 solution?
- 77.686 grams
- What is the molarity of 2.50 L of solution that contains 1.0 mol MgCl2?
- 0.4 M
- How many moles of solute are present in 400 mL of 2.0M KNO3
- 0.8 moles
Molality
molality=number of moles divided by mass of solvent in kg
- calculate the molality of a solution that has 1.5 moles added to 675 g of solvent.
- 2.222 m
Dilutions
formulation: M1 + V1 = M2 + V2
- if you dilute 70 mL of 5.0 M NaCl to make a 350.0 m L solution, what is the molarity of the dilute solution?
- M1=5.0 V1=70.0 M2=? V2=350,0 || 5.0 x 70.0 = ? x 350.0 || 350/350.0=1 || M2 = 1
- if you had 40.0 mL of a 3.00 M hydrochloric acid solution and you wanted to dilute it to 2.00 M, what would be the new volume?
- M1=3.0 V1=40.0 mL M2= 2.0M V2=? || M1 x V1=120 120/V2 --- 120/2.0=60 || New volume: 60mL
Solubility
1. Salts containing Group I elements are soluble (Li+, Na+, K+, Cs+, Rb+). Exceptions to this rule are rare. Salts containing the ammonium ion (NH4+) are also soluble.
2. Salts containing nitrate ion (NO3-) are generally soluble.
3. Salts containing Cl -, Br -, I - are generally soluble. Important exceptions to this rule are halide salts of Ag+, Pb2+, and (Hg2)2+. Thus, AgCl, PbBr2, and Hg2Cl2 are all insoluble.
4. Most silver salts are insoluble. AgNO3 and Ag(C2H3O2) are common soluble salts of silver; virtually anything else is insoluble.
5. Most sulfate salts are soluble. Important exceptions to this rule include BaSO4, PbSO4, Ag2SO4 and SrSO4 .
6. Most hydroxide salts are only slightly soluble. Hydroxide salts of Group I elements are soluble. Hydroxide salts of Group II elements (Ca, Sr, and Ba) are slightly soluble. Hydroxide salts of transition metals and Al3+ are insoluble. Thus, Fe(OH)3, Al(OH)3, Co(OH)2 are not soluble.
7. Most sulfides of transition metals are highly insoluble. Thus, CdS, FeS, ZnS, Ag2S are all insoluble. Arsenic, antimony, bismuth, and lead sulfides are also insoluble.
8. Carbonates are frequently insoluble. Group II carbonates (Ca, Sr, and Ba) are insoluble. Some other insoluble carbonates include FeCO3 and PbCO3.
9. Chromates are frequently insoluble. Examples: PbCrO4, BaCrO4
10. Phosphates are frequently insoluble. Examples: Ca3(PO4)2, Ag3PO4
11. Fluorides are frequently insoluble. Examples: BaF2, MgF2 PbF2.
2. Salts containing nitrate ion (NO3-) are generally soluble.
3. Salts containing Cl -, Br -, I - are generally soluble. Important exceptions to this rule are halide salts of Ag+, Pb2+, and (Hg2)2+. Thus, AgCl, PbBr2, and Hg2Cl2 are all insoluble.
4. Most silver salts are insoluble. AgNO3 and Ag(C2H3O2) are common soluble salts of silver; virtually anything else is insoluble.
5. Most sulfate salts are soluble. Important exceptions to this rule include BaSO4, PbSO4, Ag2SO4 and SrSO4 .
6. Most hydroxide salts are only slightly soluble. Hydroxide salts of Group I elements are soluble. Hydroxide salts of Group II elements (Ca, Sr, and Ba) are slightly soluble. Hydroxide salts of transition metals and Al3+ are insoluble. Thus, Fe(OH)3, Al(OH)3, Co(OH)2 are not soluble.
7. Most sulfides of transition metals are highly insoluble. Thus, CdS, FeS, ZnS, Ag2S are all insoluble. Arsenic, antimony, bismuth, and lead sulfides are also insoluble.
8. Carbonates are frequently insoluble. Group II carbonates (Ca, Sr, and Ba) are insoluble. Some other insoluble carbonates include FeCO3 and PbCO3.
9. Chromates are frequently insoluble. Examples: PbCrO4, BaCrO4
10. Phosphates are frequently insoluble. Examples: Ca3(PO4)2, Ag3PO4
11. Fluorides are frequently insoluble. Examples: BaF2, MgF2 PbF2.
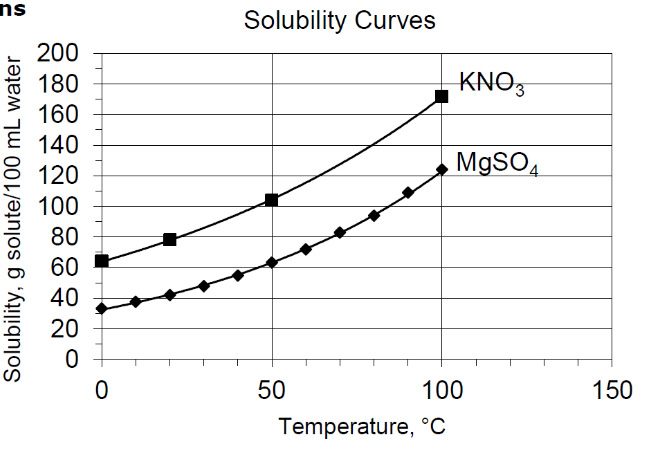
1. How much KNO3 will dissolve in 50 mL of water that is at 30 degrees Celsius?
About 42 grams of KNO3
2. If you stir 20 grams of KNO3 in 100 mL of water that is at 50 degrees Celsius, will all of it dissolve?
Yes
3. A solution that conatains 120 grams of MgSO4 in 100 mL of water at 80 degrees Celsius would be saturated, unsaturated, or supersaturated?
Supersaturated
About 42 grams of KNO3
2. If you stir 20 grams of KNO3 in 100 mL of water that is at 50 degrees Celsius, will all of it dissolve?
Yes
3. A solution that conatains 120 grams of MgSO4 in 100 mL of water at 80 degrees Celsius would be saturated, unsaturated, or supersaturated?
Supersaturated