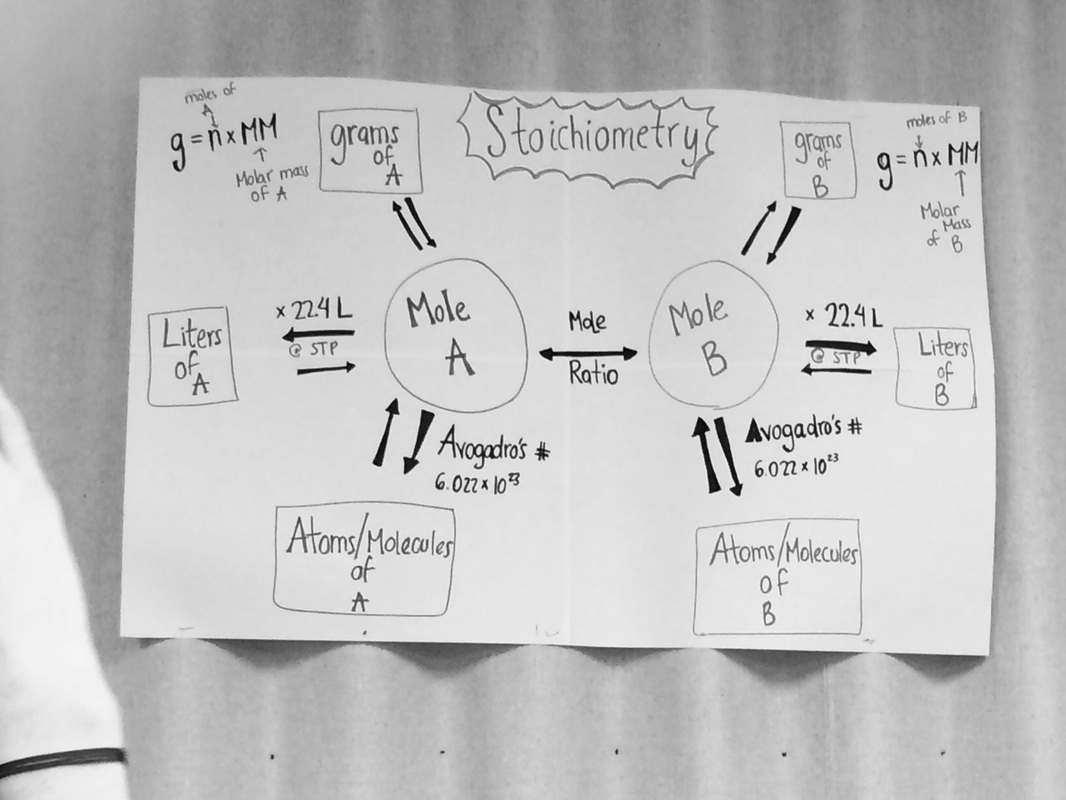
AvogAdro's Number in relation to mole conversion
1.) How is Avogadro's number related to a mole of any substance?
1 mole of a substance contains 6.02 x 10^23 particles. Avogadro's number is tha e number of atoms or molecules it takes to have one mole of a particular atom or molecule. For example, one mole of Hydrogen is just 6.02 x 10^23 atoms of hydrogen.
2.) picture below came from Ms. Del Bosque's classroom
1 mole of a substance contains 6.02 x 10^23 particles. Avogadro's number is tha e number of atoms or molecules it takes to have one mole of a particular atom or molecule. For example, one mole of Hydrogen is just 6.02 x 10^23 atoms of hydrogen.
2.) picture below came from Ms. Del Bosque's classroom
MOles to standard cooking instructions
ReciPe 1: Chocolate chip cookies
original recipe2.66 moles of flour
0.0335 moles of baking soda 0.036 moles of salt 1.084 moles of butter 0.44 moles of sugar 0.31 moles of packed brown sugar 0.624 moles of eggs 0.062 moles of vanilla 2.82 moles of chocolate chips |
Converted recipe319.5 grams of flour
2.81 grams of baking soda 2.10 grams of salt 138.9 grams of butter 77.9 grams of sugar 54.9 grams of brown sugar 100 grams of eggs 6.6 grams of vanilla extract 304.8 grams of chocolate chip |
recipe 2: Anzac Biscuits
water reduced to 0.45 moles (multiply values by 0.68)
Original Recipe 2.66 moles of flour
1.77 moles of rolled oats 4.58 moles of brown sugar 1.48 moles of shredded coconut .95 moles of almonds .96 moles of cranberries 1.19 moles of butter .89 moles of honey .63 moles of water |
Edited Recipe 1.80 moles of flour
1.208 moles of rolled oats 3.114 moles of brown sugar 1.0064 moles of shredded coconut .646 moles of almonds .653 moles of cranberries .809 moles of butter .605moles of honey .43 moles of water |
Recipe 3: Oatmeal Chocolate Cookies
water reduced to 0.5 moles (multiply values by 0.694)
Original Recipe4.26 moles of flour
0.72 moles of rolled oats 3.78 moles of butter 2 moles of eggs 0.76 moles of walnuts 2.5 moles of sugar 3.8 moles of chocolate chips 0.25 moles of baking soda 0.2 moles of vanilla |
Edited recipe2.95 moles of flour
0.5 moles of rolled oats 2.62 moles of butter 1.3 moles of eggs 0.52 moles of walnuts 1.908 moles of sugar 2.63 moles of chocolate chips 0.174 moles of baking soda 0.138 moles of vanilla |
Recipe 4: Chocolate Chip cookies
chocolate chips reduced to 2.5 moles (multiply values by 0.657)
ORIGINAL RECIPE1.06 moles of cocoa powder
3.78 moles of butter 0.76 moles of eggs 0.76 moles of walnuts 3.35 moles of sugar 3.66 moles of brown sugar 3.8 moles of chocolate chips .02 moles of baking soda .25 moles of vanilla |
EDITED RECIPE0.696 moles of cocoa powder
2.48 moles of butter 0.499 moles of eggs 0.499 moles of walnuts 2.2 moles of sugar 2.4 moles of brown sugar 2.5 moles of chocolate chips 2.4 moles of banking soda 0.164 moles of vanilla |
Recipe 5: Drop Sugar Cookies w/ Walnuts
flour reduced to 4.33 moles (multiply values by 0.812)
ORIGINAL RECIPE5.33 moles of flour
2.35 moles of butter .48 moles of eggs .96 moles of walnuts 4.19 moles of sugar .25 moles of baking soda .31 moles of vanilla |
EDITED RECIPE4.32 moles of flour
1.908 moles of butter 0.389 moles of eggs 0.779 moles of walnuts 3.4 moles of sugar .203 moles of baking soda 0.251 moles of vanilla |
Problem Solving
1. You want to make 6 grilled cheese sandwiches (use the equation below, Bd = bread and Ch = Cheese) and you have 13 slices of bread and 5 slices of cheese. How may grilled cheese sandwich can you make? And what is the limiting reagent?
2Bd + Ch→Bd2Ch
Bd = 13 Ch = 5
You have 5 slices of cheeses there for you can only have 5 grilled cheese sandwiches. Your cheese is the limiting reagent.
Problem Solving
2. You are having some friends over for dinner you want to make 8 cheese burgers, you have 8 hamburger patties, 12 bums, 7 slices of cheese, 18 slices of tomato, 16 pieces of lettuce and 9 slices of onion. Use the equation below (where buns = B, hamburger patty = H, cheese = Ch, tomatoes = T lettuce = L and onion = O) to determine how many cheese burgers you can make (what’s your theoretical yield?) and what the limiting reagent is? Hint you may need to balance the equation first.
B+ H+ Ch+ 2T+ 2L+O → BHChT2L2O
# of sandwiches per ingredient (theoretical #)
patties: 8
buns: 6
cheese:7
tomato: 9
lettuce: 8
onion:9
buns=lowest theoretical yield || limiting reagent=6
3. Balance the chemical equations below. Find the limiting reagent and the theoretical yield if there is one gram of each reagent. After each step, write a sentence or two explaining what you did.
LiOH+CO2 → LiCO3+H2O
Zn+AgNO2 → Ag+Zn(NO3)2
1. 2LiOH + CO2 --> Li2CO3 + H2O
Theoretical yield of each reagent: LiOH: molar mass 23.95
1g/ 23.95 = 0.0417 mol
output of water : ratio of water to reagent: 1:2
theoretical: 0.0208 mol
theoretical output of CO2:
molar mass: 44.01 1g/44.01=0.022 mol
ratio w water & reagent 1:1
theoretical output: 0.0227 mol
LiOH is the limiting reagent.
91.8% yield.
2. Zn + 3AgNO2 --> 3Ag + Zn(NO3)2
Zn: Molar mass (65.39)
1g/153.87 = 0.00649 mol
Ratio of Ag to AgNO2 --> 3:3 Theoretical output: 0.00649 mol
AgNO2 is the limiting reactant
Theoretical yield: 42.69%
2Bd + Ch→Bd2Ch
Bd = 13 Ch = 5
You have 5 slices of cheeses there for you can only have 5 grilled cheese sandwiches. Your cheese is the limiting reagent.
Problem Solving
2. You are having some friends over for dinner you want to make 8 cheese burgers, you have 8 hamburger patties, 12 bums, 7 slices of cheese, 18 slices of tomato, 16 pieces of lettuce and 9 slices of onion. Use the equation below (where buns = B, hamburger patty = H, cheese = Ch, tomatoes = T lettuce = L and onion = O) to determine how many cheese burgers you can make (what’s your theoretical yield?) and what the limiting reagent is? Hint you may need to balance the equation first.
B+ H+ Ch+ 2T+ 2L+O → BHChT2L2O
# of sandwiches per ingredient (theoretical #)
patties: 8
buns: 6
cheese:7
tomato: 9
lettuce: 8
onion:9
buns=lowest theoretical yield || limiting reagent=6
3. Balance the chemical equations below. Find the limiting reagent and the theoretical yield if there is one gram of each reagent. After each step, write a sentence or two explaining what you did.
LiOH+CO2 → LiCO3+H2O
Zn+AgNO2 → Ag+Zn(NO3)2
1. 2LiOH + CO2 --> Li2CO3 + H2O
Theoretical yield of each reagent: LiOH: molar mass 23.95
1g/ 23.95 = 0.0417 mol
output of water : ratio of water to reagent: 1:2
theoretical: 0.0208 mol
theoretical output of CO2:
molar mass: 44.01 1g/44.01=0.022 mol
ratio w water & reagent 1:1
theoretical output: 0.0227 mol
LiOH is the limiting reagent.
91.8% yield.
2. Zn + 3AgNO2 --> 3Ag + Zn(NO3)2
Zn: Molar mass (65.39)
1g/153.87 = 0.00649 mol
Ratio of Ag to AgNO2 --> 3:3 Theoretical output: 0.00649 mol
AgNO2 is the limiting reactant
Theoretical yield: 42.69%