types of reactions
1. Synthesis: In a synthesis reaction two or more chemical species combine to form a more complex product. For example when an egg and sperm fuse; they are 2 separate individual parts joining together to form one entity. A common example of synthesis in every day life is the synthesis of water: 2 H2(g) + O2(g) → 2 H2O(g)

2. Decomposition: Chemical decomposition, analysis or breakdown is the separation of a chemical compound into elements or simpler compounds. It is sometimes defined as the exact opposite of a chemical synthesis. Chemical decomposition is often an undesired chemical reaction. For example when a computer is dismantled for recycling; a whole is broken into components which may be used in a new process. A real life example of this is the fermentation of grapes to make wine, shown in this equation:
C6H12O6 → 2C2H6O + 2CO2
C6H12O6 → 2C2H6O + 2CO2
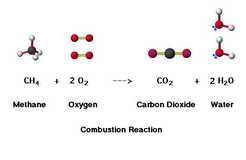
3. Combustion: A combustion reaction is a type of chemical reaction where a compound and an oxidant is reacted to produce heat and a new product. For example when people take bottles of water from a display in a store the stack of bottles is gradually being broken into pieces by the action of a series of encounters with a second item (people), creating a new kind of entity (person + water bottle). A real life example of this reaction would be the combustion of methane shown in this formula: CH4(g) + 2 O2(g) → CO2(g) + 2 H2O(g)

4. Single Replacement: A single replacement reaction is a chemical reaction where one reactant is exchanged for one ion of a second reactant. This is similar to when a couple is dancing and someone cuts in; one part of a whole is replaced to create a new entity. An example of a single replacement reaction is when one metal is sacrificed to save another metal. For example, concrete pillars have iron rebar in them for strength. However, salt water can quickly react with the iron to form iron (II) chloride. To prevent this, a metal like zinc or magnesium is attached to the rebar and will protect the iron. Here's the reaction Zn + FeCl2 -> Fe + ZnCl2

5. Double Replacement: A double replacement reaction is a chemical reaction where two reactant ionic compounds exchange ions to form two new product compounds with the same ions. This is similar to when 2 couples divorce, and then remarry each others exes; 2 unique compounds exchange components to create 2 new compounds. A real life example of double replacement reaction would be the formation of copper(II) carbonate shown in this equation: CuSO4(aq) + K2CO3(aq) ---> CuCO3(s) + K2SO4(aq)
Now I'm going to balance and classify some equations
1. NH3(g) + HCl(aq)---> NH4Cl(aq) this is a synthesis reaction and it is already balanced.
2. 8Zn(s) + S8(s) ---> 8ZnS(s) synthesis reaction; by adding 8 to the zincs there are now an even amount on both sides and the equation is balanced.
3. 8Al2S3(s) ---> 16Al(s) +3S8(s) decomposition reaction
4. H2SO4(aq) + Fe(s) ---> H2(g) + FeSO4(aq) single replacement; the equation is already balanced.
5. Mg(OH)2(aq) + H2SO4(Aq) ---> NaSO4(aq) + 2H2O(l) double replacement
1. NH3(g) + HCl(aq)---> NH4Cl(aq) this is a synthesis reaction and it is already balanced.
2. 8Zn(s) + S8(s) ---> 8ZnS(s) synthesis reaction; by adding 8 to the zincs there are now an even amount on both sides and the equation is balanced.
3. 8Al2S3(s) ---> 16Al(s) +3S8(s) decomposition reaction
4. H2SO4(aq) + Fe(s) ---> H2(g) + FeSO4(aq) single replacement; the equation is already balanced.
5. Mg(OH)2(aq) + H2SO4(Aq) ---> NaSO4(aq) + 2H2O(l) double replacement
